In pharmaceutical manufacturing and laboratory environments, precision is non-negotiable. From drug formulation and quality testing to research and development, every process depends on a controlled environment engineered to meet strict regulatory standards while supporting efficiency, safety, and repeatability.
Pharmaceutical clean room solutions and laboratory environments play a critical role in contamination control, air quality management, and regulatory compliance within pharmaceutical and laboratory facilities. As production scales, new formulations are introduced, or legacy sites are upgraded, the demand for engineered clean room solutions for pharmaceutical facilities continues to increase.
CPL Group delivers pharmaceutical and laboratory solutions designed to support compliance, protect product integrity, and optimise operational workflows across regulated environments.
The Importance of Clean Rooms in Pharmaceutical and Laboratory Environments
Pharmaceutical products are highly sensitive to airborne particulates, temperature variation, humidity, and cross-contamination. Even minor environmental deviations can result in compromised product quality, failed audits, or costly production delays.
Clean room solutions provide controlled environments where particulate levels, airflow, pressure differentials, temperature, and humidity are carefully managed. In laboratory environments, modular clean room systems support accurate testing, reproducible results, and safe handling of sensitive materials.
Key benefits of pharmaceutical clean room environments include:
- Reduced contamination risk during pharmaceutical manufacturing and laboratory testing
- Improved consistency and repeatability across regulated processes
- Enhanced protection for personnel, products, and critical equipment
- Support for validated, auditable workflows
By integrating pharmaceutical clean room systems early in facility design, operators can future-proof their sites while maintaining regulatory readiness.
Regulatory Compliance and Contamination Control in Pharmaceutical Facilities

Pharmaceutical and laboratory facilities must operate within strict regulatory frameworks, including Good Manufacturing Practice (GMP), ISO clean room classifications, and national health and safety standards.
Compliance extends beyond air quality. Facility layout, material flow, personnel movement, and controlled laboratory zones directly influence contamination risk and audit outcomes.
CPL Group’s GMP-compliant clean room design supports regulatory performance by:
- Enabling controlled airflow and pressure differentials
- Using hygienic, easy-to-clean materials
- Supporting separation of critical zones through laboratory dividers and zoning solutions
- Accommodating validation and certification requirements
These design principles allow pharmaceutical facilities to maintain audit confidence while supporting efficient daily operations.
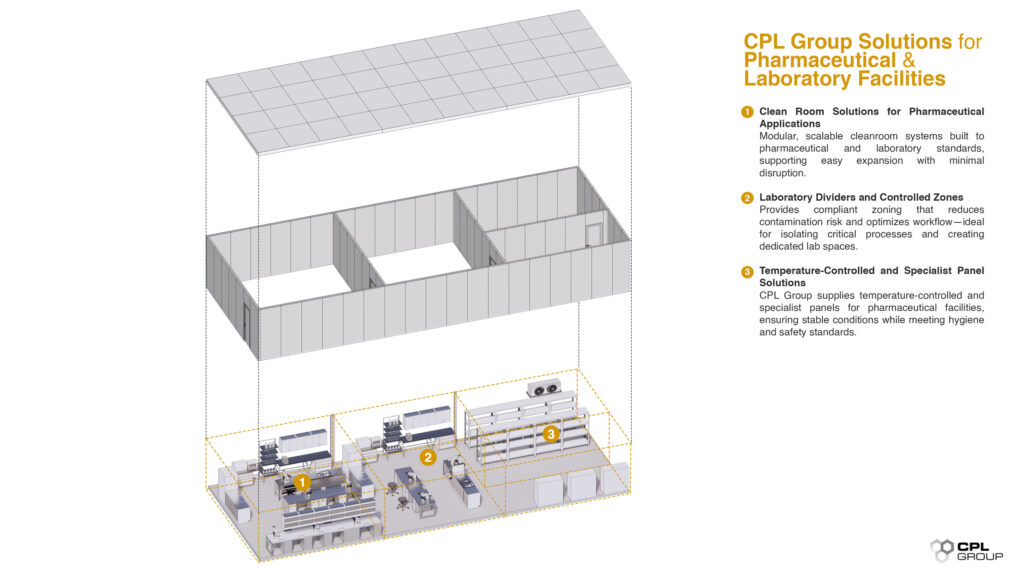
CPL Group Solutions for Pharmaceutical and Laboratory Facilities
CPL Group provides a comprehensive range of clean room and laboratory solutions for pharmaceutical environments, engineered to support compliance, safety, and operational efficiency.
Clean Room Solutions for Pharmaceutical Applications

CPL Group’s clean room solutions are modular, scalable, and engineered to meet pharmaceutical manufacturing and laboratory standards. These systems support future expansion and reconfiguration while minimising operational disruption.
Laboratory Dividers and Controlled Zones

Laboratory dividers enable effective zoning within regulated environments, helping manage contamination risks while improving workflow efficiency. These solutions are ideal for isolating sensitive processes, separating production stages, and creating dedicated laboratory zones.
Temperature-Controlled and Specialist Panel Solutions

For pharmaceutical facilities requiring cold storage or tightly controlled environments, CPL Group provides temperature-controlled panel solutions and specialist panels engineered to maintain stable conditions while supporting hygiene and safety requirements.
Each solution is designed for durability, compliance, and long-term performance in regulated pharmaceutical environments.
Improving Operational Efficiency and Long-Term ROI
Beyond compliance, purpose-built pharmaceutical clean rooms deliver measurable operational benefits. Facilities that invest in engineered clean room systems often experience:
- Reduced product loss, contamination events, and rework
- Faster manufacturing, testing, and validation cycles
- Improved staff productivity and safety
- Lower long-term maintenance and upgrade costs
Modular clean room solutions also allow pharmaceutical facilities to adapt to evolving regulations and production demands without major structural modifications.For facilities planning upgrades or expansions, CPL Group offers the opportunity to request a clean room assessment aligned with operational and compliance goals.
Why Choose CPL Group for Pharmaceutical and Laboratory Projects

CPL Group brings engineering expertise and compliance-led design to every pharmaceutical and laboratory project. From initial consultation through design, supply, and installation, solutions are developed to meet regulatory requirements while supporting operational performance.By delivering engineered solutions for regulated pharmaceutical environments, CPL Group helps operators reduce risk, improve audit outcomes, and support long-term facility resilience.
Conclusion
Clean room and laboratory environments are critical to pharmaceutical success. As regulatory expectations evolve and production complexity increases, facilities require pharmaceutical clean room solutions that deliver consistent performance, compliance, and adaptability.Learn more about CPL Group’s pharmaceutical and laboratory solutions and speak with our team to discuss your pharmaceutical facility requirements.



